|
|
| Funding for this site is provided by readers like you. | |
|
|
|
|
|||||
|
|
|||||||
|
|
|
|
| |
Science Starting To Identify the Molecular Bases of the Sense of Touch
| |
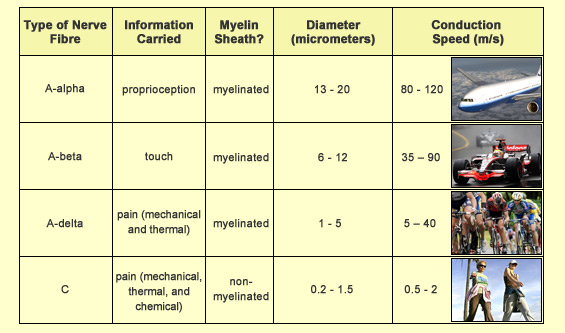
You close a door on your finger. You bump your shin on a chair. You burn your arm on the toaster. In all three cases, you experience a pain withdrawal reflex first, then an acute sensation of pain, and then a duller one. In order to understand the difference between these two types of pain—fast or acute pain and slow or dull pain—before we look at the neural pathways by which the pain signals reach the brain, we must look at where these signals start and what kind of nerve fibres they travel over. First of all, in contrast to other types of sensory fibres such as those for the sense of touch, which have specialized structures (such as Pacinian and Messner corpuscles) at their endings, nociceptive fibres (the fibres that carry pain signals) have none. Instead they have what are known as free nerve endings. These free nerve endings form dense networks with multiple branches that are regarded as nociceptors, that is, the sensory receptors for pain. These nociceptors respond only when a stimulus is strong enough to threaten the body’s integrity—in other words, when it is likely to cause an injury. There are various types of nerve fibres (axons) whose free endings form nociceptors. These fibres all connect peripheral organs to the spinal cord, but differ greatly both in diameter and in the thickness of the myelin sheath that surrounds them. Both of these traits affect the speed at which these axons conduct nerve impulses: the greater the diameter of the fibre, the thicker its myelin sheath, and the faster this fibre will conduct nerve impulses. Using these two criteria, the following types of sensory fibres can be distinguished.
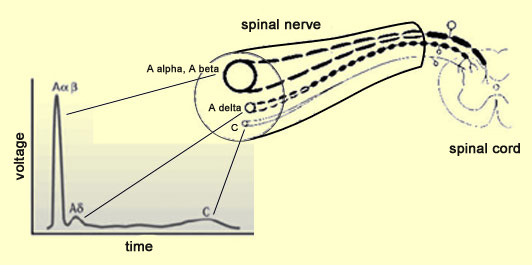
The difference between the speeds at which the two types of nociceptive nerve fibres (A delta and C) conduct nerve impulses explains why, when you are injured, you first feel a sharp, acute, specific pain, which gives way a few seconds later to a more diffuse, dull pain. This time lag is directly attributable to the difference in the conduction speeds of A delta and C fibres: their messages do not reach the brain at exactly the same time. “Fast pain”, which goes away fairly quickly, comes from the stimulation and transmission of nerve impulses over A delta fibres, while “slow pain”, which persists longer, comes from stimulation and transmission over non-myelinated C fibres. In relative terms, A delta fibres carry messages at the speed of a messenger on a bicycle, while C fibres carry them at the speed of a messenger on foot. C fibres are estimated to account for about 70% of all nociceptive fibres.
These two components of pain take different types of pathways to reach the brain: fast-pain pathways, which evolved more recently in human history, and slow-pain pathways, which evolved longer ago. The fast-pain pathways, composed of A delta fibres, also carry the signals that trigger your withdrawal reflex within a few milliseconds when you receive a painful stimulus, such as when you step on a nail.
| | |||||||||||||||||||||||||||||||||||||||||||||||||||||
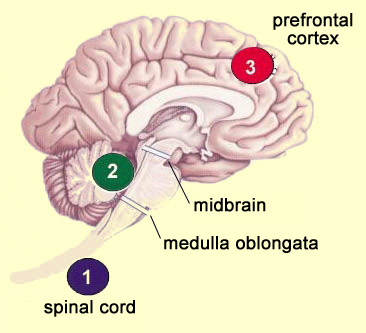
The ascending nociceptive pathways consist of A delta and C fibres that are unmyelinated or only slightly myelinated (compared with the highly myelinated tactile and proprioceptive fibres). The ascending nociceptive fibres follow several different pathways (which vary in their evolutionary age) that let the brain locate the sensation of pain and assign it an emotionally unpleasant connotation. But scientists know that these pathways are not perfect and do not always transmit pain signals intact and undistorted from the body’s periphery to the brain. The nociceptors can be highly activated without an individual’s experiencing pain—for example, when athletes or soldiers are injured or wounded but feel practically no pain in the heat of action. Or in your everyday life, haven’t you sometimes cut yourself without realizing it, because your attention was so focused on the task at hand? Or to cite another example, there is the placebo effect, where simply believing that a medication works can reduce the sensation of pain, even though the medication actually contains no active ingredients. To understand what makes all these phenomena possible, we must look at what are known as the descending pain-control pathways: neural pathways that descend from the central structures of the nervous system and diminish the pain signals travelling up the the ascending pathways from the body to the brain. Though all human perceptions are subject to varying degrees of modulation by these central structures, the power of these top-down mechanisms is greatest when it comes to controlling pain. As described above, these descending pain-control mechanisms can sometimes even completely eliminate certain forms of pain. These mechanisms thus imply a tremendous paradigm shift. They mean that pain pathways cannot be seen as direct links between the pain receptors in the body and “pain centres” in the brain. Instead, these pathways are better described in terms of concurrent ascending and descending influences—a veritable symphony of neural activity occurring simultaneously in both directions. And it is when this delicate balance tips in favour of the excitatory nociceptive messages that an individual experiences pain. Pain thus becomes less of a reflexive response to an injury and more of an “opinion” that the body forms about its physical integrity. This understanding has yielded major advances in the treatment of pain, because researchers can now seek ways to potentiate these descending pathways that inhibit pain. The theory now recognized as best describing the mechanisms involved in the descending control of pain is called the gate-control theory of pain. In this theory, the primary metaphor is that at each of the main relay points along the ascending pain pathways, there are “gates” that can be closed to make it harder for nociceptive impulses to get through. Thus, depending on how open the gates are at each of these relay points, the same level of activity in a nociceptor will not always lead to perception of the same intensity of pain.
|
| |
|
|
|
|
|
|
|
|