|
|
| Funding for this site is provided by readers like you. | |
|
|
|
|
|||||
|
|
|||||||
|
|
|
|
|
|
Donating Your Brain to Science Oligomers Help Us Keep Our Memories Junk
Food and Alzheimer’s: Closer Links Than Once Believed
|
|
Distinguishing the early stages of various dementias from normal aging is always difficult, because the first symptoms of dementias appear very gradually, often unbeknownst to the person who is experiencing them. In Alzheimer’s-type dementia, which is the most common kind, the neuronal deterioration that causes the deficits spreads through the brain in a characteristic sequence. Hence the symptoms of Alzheimer’appear in an order that, though not entirely predictable, is typical of this form of dementia. The three major stages in the progression of Alzheimer’s-type dementia are generally referred to as mild, moderate, and severe (or, alternatively, early, middle, and late). Obviously, the transition from one stage to the next is not clear-cut, and not every person will display the same symptoms in the same order, with the same severity, or for the same length of time (see box on life expectancy, below). Nevertheless, the societies that represent people with Alzheimer’s have identified some early signs and typical symptoms that appear in approximately the order that will now be described. The first part of the brain affected in Alzheimer’s is the hippocampus, which lies deep inside the temporal cortex, so it is no surprise that the first symptoms of Alzheimer’s consist of memory problems. These problems initially manifest as minor instances of forgetfulness—such as forgetting one’s keys, or an appointment, or somebody’s name—that can easily be confused with one of the common effects of normal aging.
People in this early or mild stage of Alzheimer’s preserve their oldest memories fairly well, but start to frequently forget things that they learned more recently. As they begin to ask questions more and more often, and sometimes repeatedly, about things such as an upcoming appointment or an errand that they have to run, the people around them soon start to notice. People in this stage of Alzheimer’s also use various strategies to aid their memories, such as posting notes to themselves around the house. Even so, at first, they may tend to make light of these losses of episodic memory. Other cognitive symptoms that may appear in this first stage of Alzheimer’s include a shorter attention span, difficulty in concentrating, errors in judgment, and problems with orientation in space or time. People at this stage of Alzheimer’s may, for example, tend to wander, or to get lost in places where they have been many times before. Their mood and other aspects of their behaviour start to be affected as well, sometimes resulting in exaggeration of their personality traits (angry people becoming aggressive, calm people becoming docile, and so on). These people may also experience mood swings, depression, or nervousness. Occasional awareness of the deterioration of their faculties can become a source of anxiety. People in the early stage of Alzheimer’s may also have slight problems with motor co-ordination, but in general can continue to live relatively independent lives with a minimum of assistance. People are considered to have entered the moderate stage of Alzheimer’s-type dementia when their judgment starts to be so impaired that it becomes hard for them to structure their daily activities. This is the stage when people lose their independence. For example, they may have trouble in completing familiar tasks that they have been performing for their entire adult lives, such as preparing their meals, grooming themselves, or budgeting their money. Their cognitive faculties, such as memory, continue to deteriorate, and they find it harder and harder to learn and retain new information, but they still retain their memories of their youth and middle ages. In the moderate stage of Alzheimer’s, amyloid plaques and neurofibrillary tangles gradually extend into the parts of the brain involved in perceiving one’s body separately from the objects around it, as well as the parts involved in language. That is why people begin to experience difficulties not only in their daily tasks, but also in their social and work lives, which suffer tremendously from the deterioration in their language abilities. For people at this stage of Alzheimer’s, the slightest conversation becomes hard work. They lose the thread of their ideas, can no longer find the words to express themselves, and do not understand clearly when things are explained to them. At this same time, they also stop reading and writing. As moderate Alzheimer’s progresses toward the severe stage, it is not uncommon for people to develop more severe, disabling problems, such as insomnia, fluctuations in appetite, and changes in personality. that make their lives even more difficult. Once Alzheimer’s-type dementia reaches the severe stage, most of the cortex is seriously damaged. People have great difficulty in expressing themselves and in recognizing their friends and even their close family. Mental confusion, aggravated by severe memory loss, often leads to hallucinations or paranoid delusions. Behaviourally, people with severe Alzheimer’s often become irritable or apathetic, make stereotyped or inappropriate movements, laugh or cry for no apparent reason, and so on. However, two people both of whom are assessed as being at the severe stage of Alzheimer’s may manifest it in different ways. In one, the cognitive faculties may be more affected, while in the other, it may be the ability to perform daily activities independently. In the severe stage of Alzheimer’s, people often neglect their personal hygiene, may forget to eat, and lose bladder and bowel control. At this stage, they need to be constantly supervised either by family members or in a long-term residential care centre. The most important objective at this stage is to support the person who has Alzheimer’s to give them the best possible quality of life. Over time, Alzheimer’s-type dementia often evolves into a “mixed dementia"—in other words, it co-exists with other forms of dementia, such as vascular dementia and Lewy body dementia. At any stage in the progression of dementia, the individual can die from some other disease. In the late, severe stage of Alzheimer’s-type dementia, many deaths result from secondary infections, often pneumonia caused by saliva or food that the person accidentally inhales because he or she has trouble in swallowing.
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Junk Food and Alzheimer’s: Closer Links Than Once Believed
|
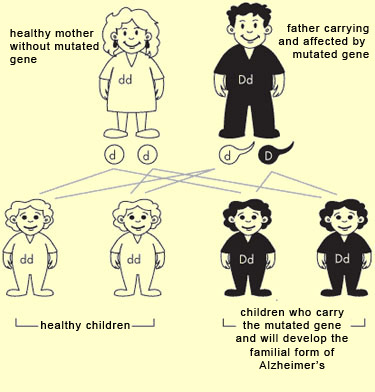
Although research has contributed greatly to our knowledge of the mechanisms at work in Alzheimer’s-type dementia, its ultimate causes remain unknown. But science has uncovered a number of environmental and genetic risk factors for Alzheimer’s (see sidebar). As in other chronic conditions, these risk factors may combine to trigger this dementia. But before we consider these risk factors, we should note that there are two forms of Alzheimer’s: familial and sporadic. The familial form tends to have an early onset, with deficits appearing between ages 30 and 65, whereas the sporadic form usually develops after age 65. Aside from this difference, however, the two forms are entirely similar as regards the kinds of symptoms involved and the biochemical and physical abnormalities observed in the brain. The main distinction is in their relative frequency. The sporadic form accounts for the vast majority of all cases, while the familial form is far rarer, accounting for only about 5 to 7%. Also, the sporadic form has no decisive genetic component, whereas the familial form is heavily determined by certain genes. When we talk about risk factors for Alzheimer’s, we must therefore first specify which of these two forms we are talking about, because the genetic risk factors for the two forms are very different. Whether a person will develop the familial form of Alzheimer’s is largely determined by his or her genes. Researchers have identified three different genes that are involved in this form of Alzheimer’s, on three different chromosomes: the gene for the amyloid precursor protein (APP), on chromosome 21; the presenilin 1 (PS1) gene, on chromosome 14; and the presenilin 2 (PS2) gene, on chromosome 1. If a person inherits any one of these genes when it has been rendered defective by a mutation, that person will develop the familial form of Alzheimer’s. These mutated genes will in turn produce defective proteins, and researchers have found several signs that these dysfunctions lead to an increase in a form of beta-amyloid protein that accumulates in toxic plaques in the brain. Familial Alzheimer’s is classified as an autosomal dominant disorder: “autosomal” because the genes in question are not located on an X or Y sex chromosome (which determines the person’s sex), and “dominant” because a person needs to inherit only one copy of the mutated gene, from one parent, in order to develop the disorder. Thus someone who inherits one of these mutated genes will always develop Alzheimer’s and pass it on to half of his or her descendants. In other words, a child who has one parent with the family form of Alzheimer’s will have a 50% chance of inheriting it as well.
That said, only about half of all early-onset cases of Alzheimer’s are due to familial-form mutations. And even if several members of the same family develop Alzheimer’s disease, that does not necessarily mean that it is the familial form. It could be be the sporadic form, which is not entirely independent of family history. As scientists now know, even for the sporadic form of Alzheimer’s, having a close relative (father, mother, brother, or sister) with this kind of dementia increases the risk of developing it oneself. Thus, there are genetic risk factors for the sporadic form—genes that do not cause Alzheimer’s directly, like the genes for the familial form do, but that are more common in people who have sporadic Alzheimer’s (even though these genes are sometimes also found in people who do not). A number of genes, such as CLU and CR1, are suspected of being risk factors for the sporadic form of Alzheimer’s disease, but the best known is unquestionably the type 4 allele of the gene for apolipoprotein E (ApoE4), discovered on chromosome 19 in 1993. This ApoE gene contributes to the synthesis of a protein that helps to transport cholesterol in the body, and various explanations have been offered for its harmful effects in Alzheimer’s. The ApoE gene has three main alleles—ApoE2, ApoE3, and ApoE4. Unlike the genes that cause familial Alzheimer’s, these three alleles are not harmful mutations that result in dysfunctional proteins, but they are three different versions of the gene that have appeared in the course of evolution and that vary in their effects. These three variants of the ApoE gene occur in known proportions in the population: 51% for ApoE2, 80% for ApoE3, and 15% for ApoE4. The most common of the three, ApoE3, has no effect on the incidence of Alzheimer’s disease. ApoE2 might actually reduce the incidence of Alzheimer’s in people who carry it. It is the people who carry the ApoE4 gene who are the most likely to develop Alzheimer’s. Since each of us carries two copies of every gene (one from our mother and one from our father), people who inherit only one copy of ApoE4 have about a three times higher risk of Alzheimer’s. But for people who carry two copies of ApoE4, the risk is about 11 times higher. This means that at age 65, about half of the latter group will have Alzheimer’s-type dementia. Thus the presence of the ApoE4 allele is neither necessary nor sufficient for developing Alzheimer’s, but the risk increases with the number of copies of this allele. Nor does this mean that someone who carries two copies of the ApoE4 allele will necessarily develop Alzheimer’s, but it does mean that someone who does not carry any copies of this allele may nevertheless develop Alzheimer’s. Both for people who carry the ApoE4 gene and for people who do not, the other major risk factor for the sporadic form of Alzheimer’s is simply age. Indeed, age is considered the greatest risk factor for developing Alzheimer’s, because even though this type of dementia can appear before age 50, most of the people who have it are over age 65. One thing is certain: there are no teenagers who have Alzheimer’s. Apparently, as people age, the brain’s natural repair mechanisms become less effective, though the extent of this loss can vary from one individual to another. Other factors for which the risk is known to increase with age, such as high cholesterol and overweight, are also recognized as risk factors for Alzheimer’s. So is being female: after age 65, two times more women than men have Alzheimer’s. Because women live longer than men, on average, and because age is a risk factor for Alzheimer’s, this greater longevity among women most likely explains at least part of this observed difference. Moreover, the longer women live, the more likely they are to develop diseases whose prevalence increases with age, such as diabetes, which is also a risk factor for Alzheimer’s. Another proposed explanation for women’s higher risk of Alzheimer’s is that the female hormone estrogen has protective effects on the brain, and the levels of this hormone drop in women after menopause. Lastly, in early 2009, a study found, for the first time, a gene that may increase women’s risk of Alzheimer’s. It is an abnormal variant of the gene for the protein PCDH11X and occurs on the X sex chromosome, of which women have two copies and men have only one (in men, the other chromosome in the pair is the Y chromosome, inherited from their fathers). This study showed that only those women who have this abnormal PCDH11X gene on both of their X chromosomes have a higher risk of developing Alzheimer’s. Many other risk factors for Alzheimer’s have been identified. For example, the risk factors for cardiovascular disease are closely related to those for Alzheimer’s: high blood pressure or high cholesterol can trigger or accelerate the progression of Alzheimer’s. In fact, there is evidence that giving people effective treatment to control their high blood pressure or high cholesterol reduces the risk of their developing Alzheimer’s, if they do not already have it, and slows its progress, if they do. The discovery that a defective version of the SORL1 gene seems to be involved in some cases of Alzheimer’s makes the case for this connection even stronger, because this gene is also related to cholesterol. This variant of the SORL1 gene manufactures less of the receptors for the ApoE protein, which is also clearly associated with cholesterol metabolism and with the amyloid plaques seen in Alzheimer’s. As mentioned above, diabetes (specifically, type 2, “adult” diabetes) is another risk factor for Alzheimer’s, in particular because of the cardiovascular problems associated with diabetes. It is also known that, just as people with diabetes do not assimilate glucose well into their bodies, people with Alzheimer’s do not assimilate glucose well in their brains. Thus, though people with Alzheimer’s may not be diabetic in the usual sense of the term, they do seem to produce less insulin in their brains (a process independent of the production of insulin elsewhere in the body), and their neurons seem to be less sensitive to the insulin that their brains do produce. Stress is a risk factor for Alzheimer’s, because it increases the secretion of the hormone cortisol, whose harmful effects on populations of neurons are well known. Another way that stress increases the risk of Alzheimer’s is by increasing the production of free radicals that accelerate aging of brain tissue, when age is such an important risk factor for Alzheimer’s. Because of the close connections between chronic stress, behavioural inhibition, and depression, depression is often associated with the risk factors for Alzheimer’s. Strokes, including “mini-strokes", can foster the earlier onset not only of vascular dementias but of Alzheimer’s-type dementia as well. The same goes for serious head injuries, often accompanied by loss of consciousness (the repeated concussions suffered by boxers, for examples). Studies have shown that people who have completed less than six years of school seem to be more susceptible to Alzheimer’s disease. But it might also be that some factors associated with limited education, such as low income resulting in an unhealthy lifestyle, may explain this greater susceptibility. More generally, lack of intellectual stimulation and lack of physical activity have an aggravating effect on Alzheimer’s, as on many other pathologies. |
| |
|
|
|
|
|
|
|
|